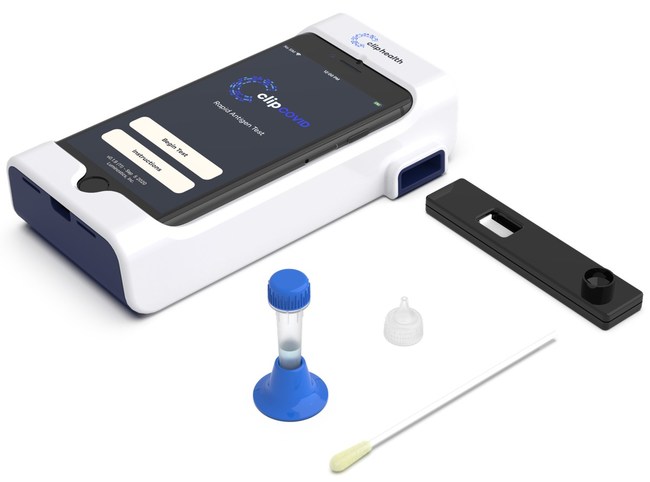
BOZEMAN, Mont., April 28, 2021 /PRNewswire/ — Alercell, announced today that it will begin distributing the Clip COVID Rapid Antigen Test, a revolutionary antigen analyzer along with its antigen COVID-19 test collection kits, across the U.S through its network and partnerships. The Luminostics Clip Analyzer is an analyzer intended to be used for objective readout of cartridge-based immunoluminescent in vitro diagnostic assays manufactured by Luminostics. The Clip COVID Rapid Antigen Test received FDA Emergency Use Authorization in December 2020 for point-of-care use in CLIA laboratories including CLIA Waived Laboratories.
Antigen tests are diagnostic tests that checks to see if you are infected with the coronavirus. The test looks for proteins (antigens) in a sample taken from your nose or throat. Antigen testing is designed to be quicker and simpler than other tests for the coronavirus and could be conducted at the point of care. A breakthrough in coronavirus antigen testing could become part of the broader solution for the widespread testing shortages. According to the Centers for Disease Control and Prevention (CDC), antigen tests can be used in a variety of testing strategies to respond to the COVID-19 pandemic and are helpful in determining whether a person diagnosed with COVID-19 remains infectious.
Such tests need to have high accuracy and “we are pleased to work with Luminostics because of the very high Sensitivity and Specificity of the Clip COVID Rapid Antigen Test. We believe that Luminostics technology is ahead of the competition. This new high-sensitivity antigen test is another example of Alercell ‘s commitment to providing people with the information they need to make important health decisions,” Said Dr. Alexandre Scheer Chief Medical Officer of Alercell.
The Clip COVID test’s hardware and software leverage patented innovations in nanotechnology, materials chemistry, and signal processing to enable high-accuracy, cost-effective diagnostics. The Luminostics core innovation is a new type of nanoparticle that is very sensitively detectable using a smartphone’s built-in camera and flash in combination with Luminostics’ proprietary hardware and software. Because of its 21st century technology the system allows automated reporting results linkable to LIMS & EHR systems and reportable to public health authorities. The system is efficient with a single operator able to run tens of tests each hour with a hands-on time of only 90 seconds per test. At the difference of standard Antigen tests, the ClipCovid is a machine read Antigen allowing much higher accuracy than the competition.
Alercell continues to recommend that individuals follow health guidelines, including wearing a mask in public, socially distancing, frequently washing hands and avoiding large groups, and receiving a COVID-19 vaccine as availability increases and the CDC guidance expands to more eligible groups.
ABOUT ALERCELL
Alercell is a Biotechnology company specialized in infectious diseases, based in Bozeman, Montana with a satellite office in New York City. Created by a group of veterans of medical research, Alercell’s core business is in the discovery, development, distribution, manufacture, and sale of efficient treatment for infectious diseases including the Coronavirus and cancer prevention diagnostic testing. For more information, please visit:www.alercell.com
ABOUT LUMINOSTICS
Luminostics is a California-based diagnostics company with a mission to increase healthcare accessibility and efficiency by designing, developing, manufacturing, and delivering products that enable affordable, widespread access to actionable health testing with immediate follow-up. The company’s smartphone-based rapid tests — beginning with the Clip COVID Rapid Antigen Test — offer digitally connected, highly accurate, and affordable testing or monitoring solutions for various types of pathogens, proteins, hormones, and other biomolecules in point-of-care, enterprise, and at-home settings. For more information, please visit: https://luminostics.com/
Forward Looking Statements
Certain statements made in this release are “forward looking statements” within the meaning of the “safe harbor” provisions of the United States Private Securities Litigation Reform Act of 1995. When used in this press release, the words “estimates,” “projected,” “expects,” “anticipates,” “forecasts,” “plans,” “intends,” “believes,” “seeks,” “may,” “will,” “should,” “future,” “propose” and variations of these words or similar expressions (or the negative versions of such words or expressions) are intended to identify forward-looking statements. These forward-looking statements are not guarantees of future performance, conditions or results and involve a number of known and unknown risks, uncertainties, assumptions and other important factors, many of which are outside Alercell ‘s control, that could cause actual results or outcomes to differ materially from those discussed in the forward-looking statements. Important factors, among others, that may affect actual results or outcomes include the inability to complete the proposed business combination or the Extension; the inability to recognize the anticipated benefits of the proposed business combination; costs related to any business combination; Alercell’s ability to manage growth; Alercell’s ability to execute its business plan; the timing of revenues from existing customers, including uncertainties related to the ability of Alercell’s customers to commercialize their products and the ultimate market acceptance of these products; the uncertain impact of the COVID-19 pandemic on Alercell’s and its customers’ businesses; uncertainties related to Alercell’s estimates of the size of the markets for its products; the rate and degree of market acceptance of Alercell ‘s products; the success of other competing products and services that exist or may become available; Alercell’s ability to identify and integrate acquisitions; rising costs adversely affecting Alercell’s profitability; uncertainties related to Alercell’s potential litigation involving Alercell or the validity or enforceability of Alercell’s intellectual property; and general economic and market conditions impacting demand for Alercell’s products and services. Other factors include the possibility that any proposed business combination does not close, including due to the failure to receive required security holder approvals, or the failure of other closing conditions. Alercell does not undertake any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
Disclaimer
This release shall neither constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any jurisdiction in which the offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction.
Media Contact
Alercell, Inc.
Mr. Frederic Scheer, CEO
Email:
308638@email4pr.com
Dr. Alexandre Scheer, Chief Medical Officer
Email:
308638@email4pr.com
Tel: (406) 233-6434
Luminostics, Inc.
308638@email4pr.com
Tel (760) 709-2230
SOURCE Alercell, Inc.